Latin Name
Common Name
Found 66 Resources
Page 1 of 1
Page 1 of 1
Asian Spongy Moth
Lymantria dispar
Cactus Moth
Cactoblastis cactorum
European Larch Canker
Lachnellula (Dasyscypha)
European Oak Bark Beetle
Scolytus intricatus
European Spongy Moth
Lymantria dispar
Fusarium euwallacea
Fusarium euwallacea
Harrisia Cactus Mealybug
Hypogeococcus pungens
Kuroshio Shot Hole Borer
Euwallacea kuroshio
Oak Dieback
Phytophthora quercina
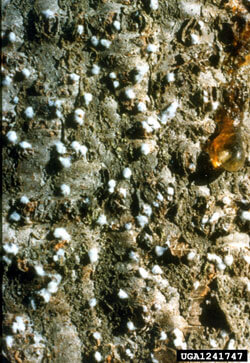
Oystershell scale
Lepidosaphes ulmi
Pine Flat Bug
Aradus cinnamomeus